Trial Design
Trial Design
Statistical approaches to trial design in early clinical development, with a focus on biomarker-driven and adaptive methods.
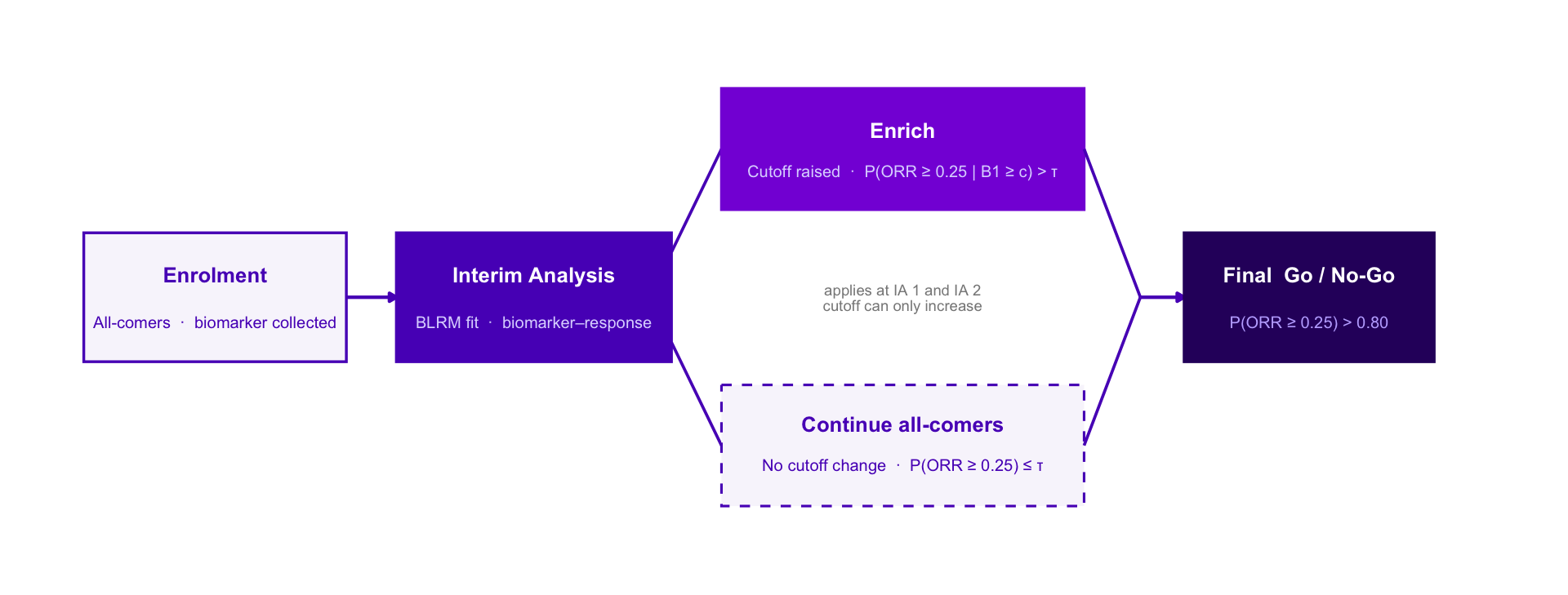
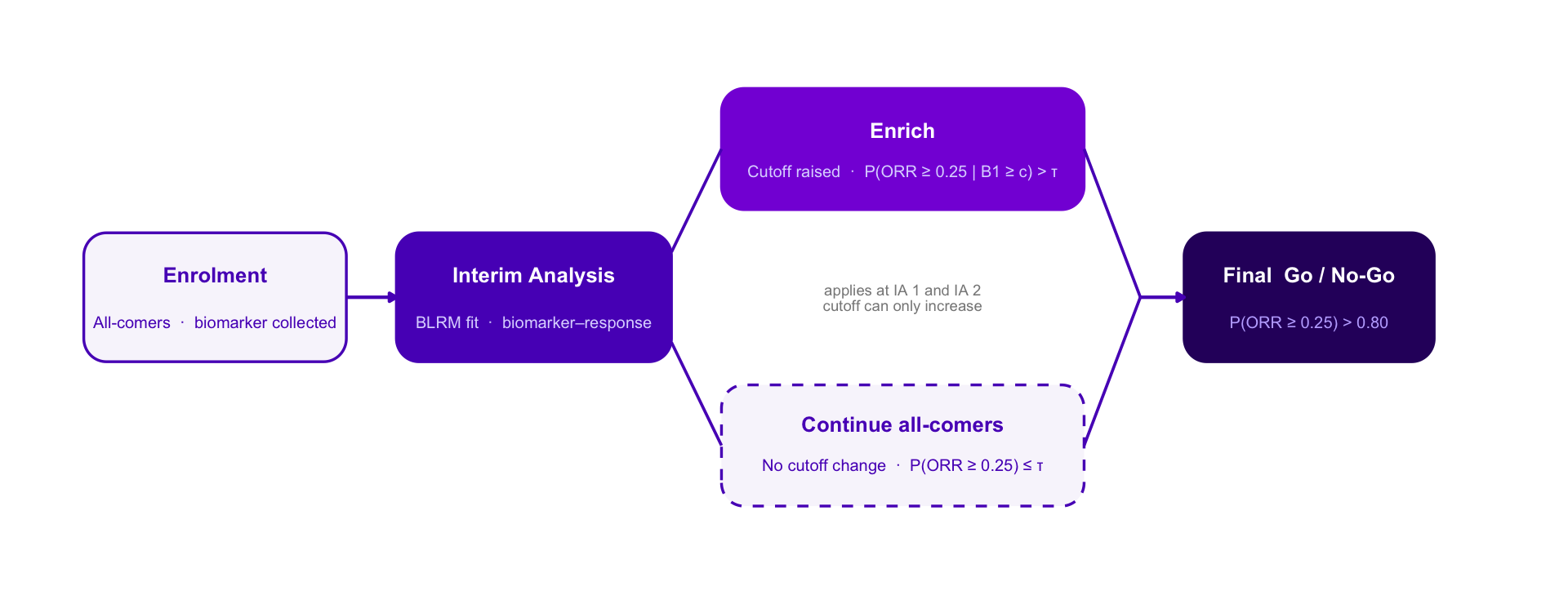
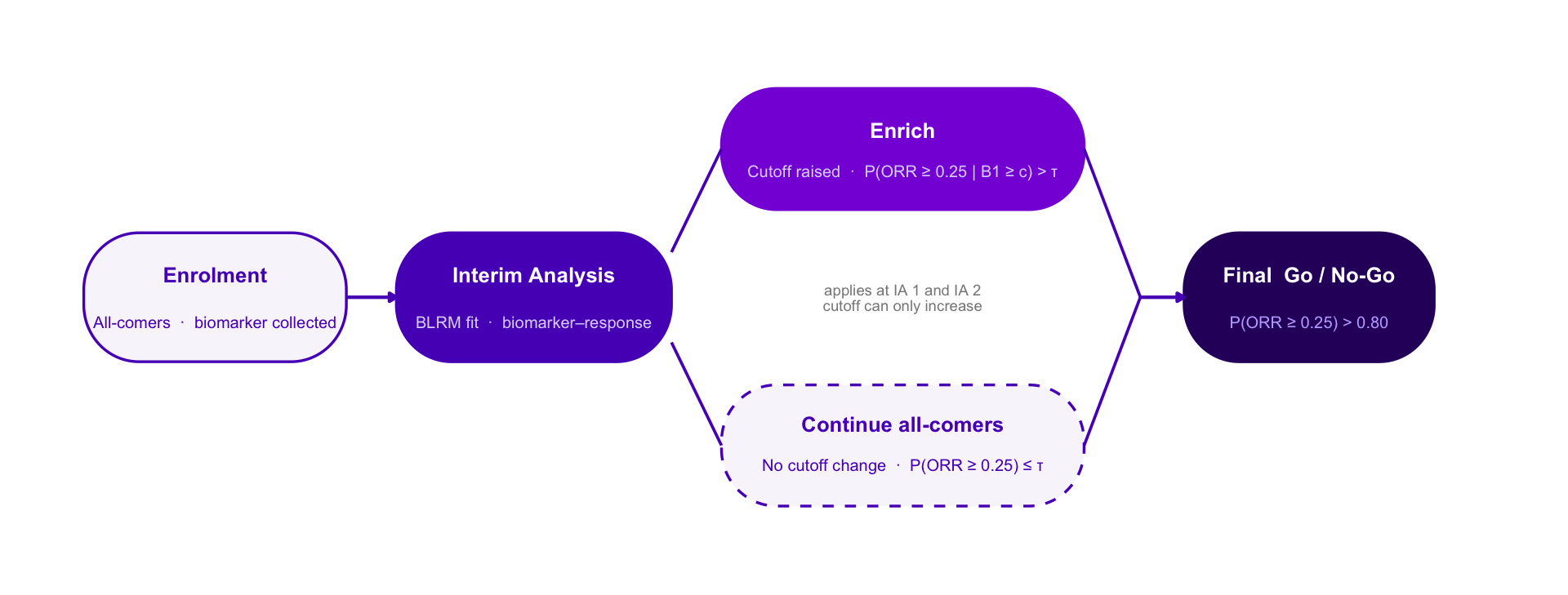
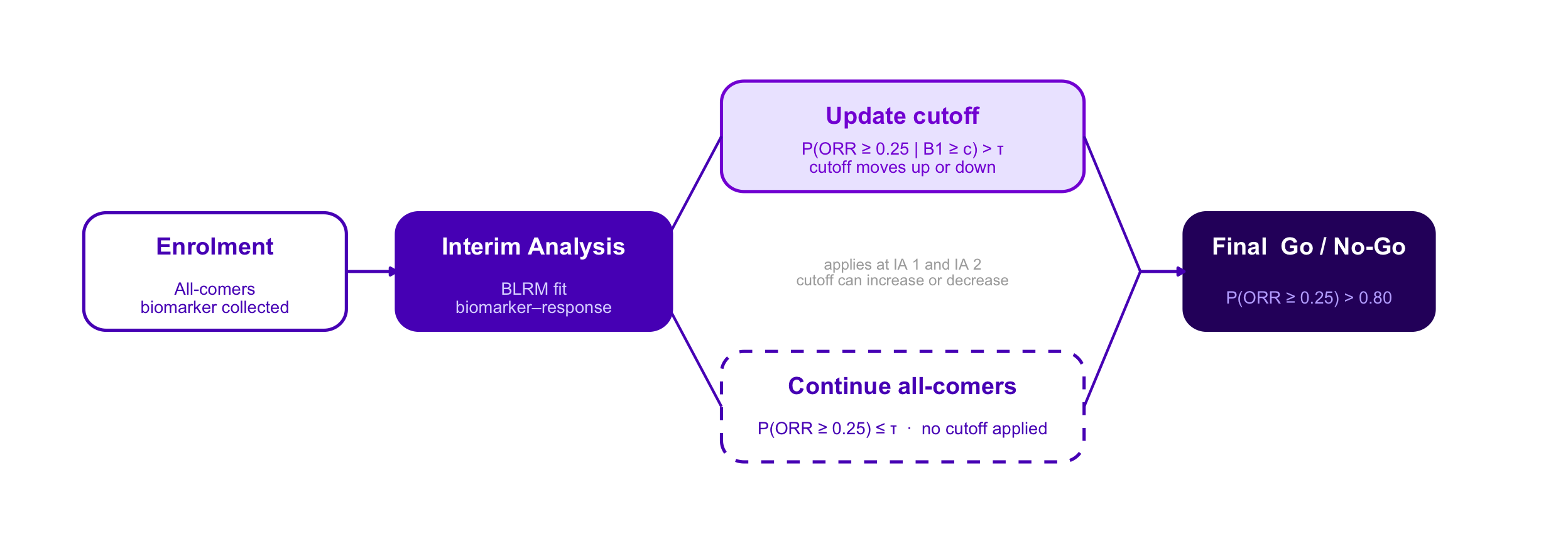
Adaptive Enrichment — Option C Style Variants
Same design throughout: single-arm Ph2, n = 300, IAs at n = 100 and n = 200, BLRM-driven biomarker cutoff, ratchet rule, final Go / No-Go at P(ORR ≥ 0.25) > 0.80.