Bio.dy/dx
Biostats insight, opinion and tools for clinical development.
Trial design, analysis, biomarkers, and simulation in R. By Paul Steven.
Practical insights in clinical trial design, complex endpoints, and biomarker-driven development — drawn from 13+ years across early phase oncology, rare disease, IVDs, and companion diagnostics.
Expertise
Trial Design
Bayesian adaptive designs for early phase trials — dose escalation (BOIN), dose optimisation (MATS), basket trials, and adaptive enrichment. IVD and analytical studies utilizing device endpoints.
Complex Endpoints
Non-standard and translational endpoints including PD, ctDNA, MRD, and analytical device output. Statistical strategy aligned with underlying science and regulatory expectations.
Simulation in SAS and R
Simulation frameworks for dose-response, biomarker thresholds, and design operating characteristics. R/Shiny tools for interactive exploration of Bayesian models.
Latest Insights
19 Mar 26
Adaptive Enrichment Explorer
Interactive R/Shiny app for simulating adaptive enrichment in biomarker-driven oncology trials.
13 Mar 26
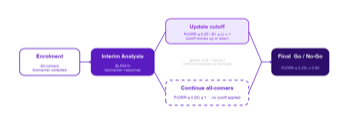
Adaptive Enrichment in Biomarker-driven Oncology
A practical explainer of adaptive enrichment in biomarker-driven oncology, illustrated with a single-arm Bayesian phase 2 example.